Pls send inquiry to info@nbinno.com if you have any interests, thank you!
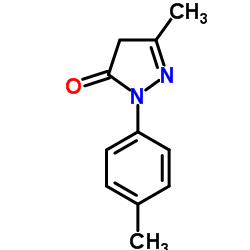
Related News: The rigosertib Pre-approval Access Program is expected to launch in first half of 2020 and will allow Inceptua to supply intravenous rigosertib within designated countries, primarily and initially concentrated in selected countries in Europe, in response to physician requests for patients with higher-risk MDS who have exhausted all available treatment options, and are not eligible for or have no access to the INSPIRE study.2,7-Dibromo-9,9-Spiro-Bifluorene CAS:171408-84-7 At ASH, scientists from the Company and MSK presented new in vivo preclinical data demonstrating that FT819 exhibits durable tumor control and extended survival.Benzotriazol-1-yloxytris(dimethylamino)phosphonium Hexafluorophosphate Rigosertib, in its intravenous formulation, is currently in Phase 3 clinical development for the treatment of higher-risk MDS.6-Bromo-1-hexanol In these situations, the API is not a single substance but the culmination of various ingredients.At ASH, scientists from the Company and MSK presented new in vivo preclinical data demonstrating that FT819 exhibits durable tumor control and extended survival.


